Karen Lusky
January 2019—Pathologists who aren’t microbiologists can provide a diagnosis of parasitic disease if they take into account parasite life cycles and tissue tropisms. Julie A. Ribes, MD, PhD, made that key point in cases she presented in her CAP18 session, “Update on Invasive Parasitic Infections for Surgical Pathologists.” Dr. Ribes added learning material to most of the cases, she said, but the cases come from parasites she has seen and known in her own professional life.
 “Tropism is the organism’s predilection to home in on specific tissues,” Dr. Ribes, director of clinical microbiology at UK HealthCare in Lexington, Ky., told CAP TODAY in a recent interview. “We can predict potentially what organism is present there by looking at what tissue they have decided to take up residence in.” In lung disease, for example, “you know there are relatively few organisms that are going to have any type of transit time through the lung.” The pathologist is therefore going to focus on those organisms likely to pass through the lung or to localize there indefinitely. “Once you have in mind what organisms are likely to be in a particular tissue,” Dr. Ribes said, “you can then correlate with the parasitic structures identified to home in on a definitive diagnosis.”
“Tropism is the organism’s predilection to home in on specific tissues,” Dr. Ribes, director of clinical microbiology at UK HealthCare in Lexington, Ky., told CAP TODAY in a recent interview. “We can predict potentially what organism is present there by looking at what tissue they have decided to take up residence in.” In lung disease, for example, “you know there are relatively few organisms that are going to have any type of transit time through the lung.” The pathologist is therefore going to focus on those organisms likely to pass through the lung or to localize there indefinitely. “Once you have in mind what organisms are likely to be in a particular tissue,” Dr. Ribes said, “you can then correlate with the parasitic structures identified to home in on a definitive diagnosis.”
In her talk, Dr. Ribes provided other takeaways from her cases: Measure up, both worms and eggs. Formulate a differential based on the tissue location and stages of the parasite seen. Not all parasitic infections are accompanied by eosinophilia. Being aware of a parasite’s life cycle can be helpful. “Many of these organisms have relatively complex life cycles, so the particular stage that the human can actually serve as the host is going to be restricted,” she explains.
Dr. Ribes presented the case of a 64-year-old Vietnamese patient who had lived in the Commonwealth of Kentucky for decades. (The two “buzzwords,” she hinted, are Vietnamese and the Commonwealth of Kentucky.) The patient had undergone a bone marrow transplant for non-Hodgkin lymphoma and was taking prednisone and CellCept to support the transplant. After a prednisone taper, he was evaluated for weakness and diarrhea. “An endoscopic biopsy of the patient’s duodenum was taken when an ulcerative lesion was identified during the endoscopic workup,” Dr. Ribes said.
The biopsy specimen revealed “large, thin worms at 10× hanging out in the mucosal of the crypts.” In more slices through the collected tissue, they could see other structures at 50×. Those resembled “large owl-eye-like inclusions, again, retained within the bowel wall,” Dr. Ribes said. In examining further, these structures varied considerably in size. Most of the interior components were “relatively amorphous.”

Showing an image of the specimen, Dr. Ribes pointed out that the circled structure at the top is “a very different size distribution than the rest of them.” (Fig. 1). These represented slices through eggs at different levels, giving the impression they varied in size. “It’s not that we have a huge variation in size in these structures, but that we just cut in a different place.” The larger ones are slices in the middle, and the smaller ones represent slices at the tapered ends of the eggs.
An image of another slice from the specimen displayed a “haloed structure” that Dr. Ribes dubbed “egg in tissue” on the left (Fig. 2). It was slightly larger than the image on the right, which is a hookworm egg, as it would be seen in stool. (The hookworm wasn’t from the patient.) The hookworm egg interior contents are “globular in formation,” she said, “so this is an unembryonated egg.” By contrast, the “egg in tissue” appears to have “lost the globular component and is heading toward becoming a larva.”
First of two parts. Next month: Schistosomiasis, Naegleria fowleri, maggots
In looking at the specimen further, they could see that some of the eggs were completely embryonated. “We have actual larvae that are mature and ready to erupt from these eggs. These eggs are going through their full maturation and then hatching in situ without being shed in the stool. So unlike that hookworm egg that we saw before, we are not expecting to see these eggs in a stool specimen.” (Fig. 3).
 “This is strongyloidiasis,” Dr. Ribes said, noting the man was from Vietnam, “a perfect place” to get strongyloidiasis. And for decades, he has been in the Commonwealth of Kentucky. “Another perfect place to pick up strongyloidiasis. It is our Commonwealth worm.”
“This is strongyloidiasis,” Dr. Ribes said, noting the man was from Vietnam, “a perfect place” to get strongyloidiasis. And for decades, he has been in the Commonwealth of Kentucky. “Another perfect place to pick up strongyloidiasis. It is our Commonwealth worm.”
Dr. Ribes pointed out that Strongyloides has two different types of larvae (Fig. 4). The cycle begins with “free-living adult worms mating in the environment, producing eggs that produce rhabditiform larvae.” These can mature back into adults and continue that life cycle in the environment.
“If, however, they go on to mature into a filariform, free-living infective stage, they are prepared to become the parasite for an unsuspecting human walking barefoot in the dew-laden grass of eastern Kentucky,” she said. The filariform larvae will “percutaneously invade” and obtain access to the venous system. “They will be impacted into the patient’s lung, be coughed up, swallowed down, and then will take up residence in the duodenum, where the adult worms will then go ahead and produce more eggs.”
Only the females are invasive. “There is no mating that occurs in the host.” Reproduction occurs by parthenogenesis, Dr. Ribes said, which is the maturation of an unfertilized egg.
The patient from Vietnam is likely to have had a chronic infection from Vietnam, Dr. Ribes said. “When we throw steroids at this infection, what we see is that rhabditiform larvae are stimulated to mature directly into the filariform larvae, which is then capable of directly penetrating the skin or bowel wall.”
In the setting of high-dose steroids, or even low-dose, “these guys will enhance that hyperinfection syndrome, and we can see infections with massive numbers of organism that will be produced.” The filariform larvae “may drag with flora from the bowel as they invade tissue and vasculature,” she said, “and you can see sepsis and fungemia, or meningitis.” The filariform larvae can be found in various tissues.
 The Vietnamese patient from Kentucky had strongyloidiasis and hyperinfection syndrome. “When we looked at this stool, it was full of rhabditiform larvae,” Dr. Ribes said. “They began treating him, and the next day you could actually see these adult worms being shed in the stool,” along with rhabditiform and filariform larvae. “He was shedding huge numbers of these in his stool over the next several days.”
The Vietnamese patient from Kentucky had strongyloidiasis and hyperinfection syndrome. “When we looked at this stool, it was full of rhabditiform larvae,” Dr. Ribes said. “They began treating him, and the next day you could actually see these adult worms being shed in the stool,” along with rhabditiform and filariform larvae. “He was shedding huge numbers of these in his stool over the next several days.”
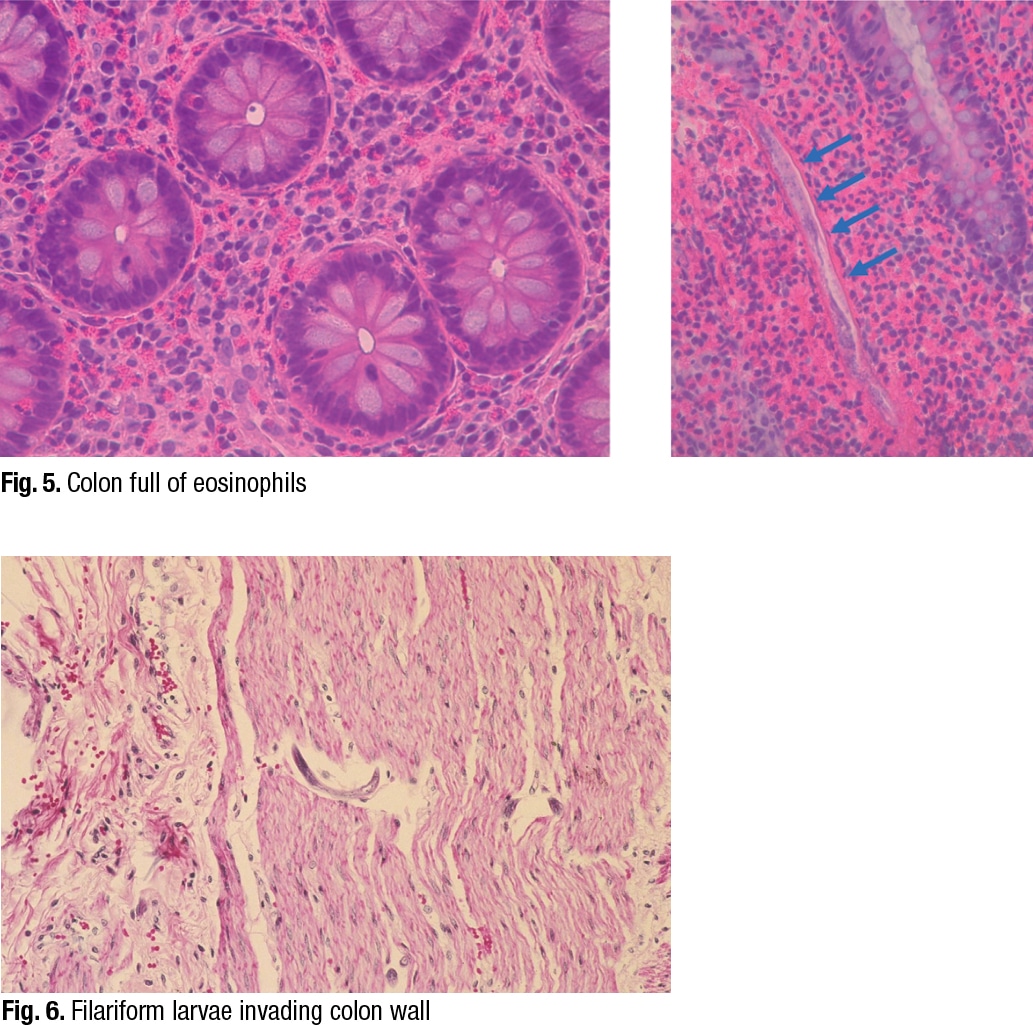
In reviewing their past cases of strongyloidiasis, Dr. Ribes and colleagues found that eosinophilia was probably present in about 20 to 25 percent of them, and it was intermittent. “Depending on the patient, eosinophilia varies greatly,” she said, showing an image of eosinophils in the colon (Fig. 5). On the right side of the image is another characteristic feature. “As the rhabditiform larvae are being stimulated by steroids to mature into filariform free-living invasive forms, we can see that they may begin piercing through the bowel wall and be identifiable as what I refer to as speckled bands.”
She pointed to a different case (Fig. 6). “In this case there’s no eosinophilia,” she said, “but notice speckled bands. We have a hyperinfection syndrome even here, but without all that eosinophilia. So the eosinophilia may be helpful when present, but not necessarily helpful if not present, but certainly be looking for the speckled bands.”
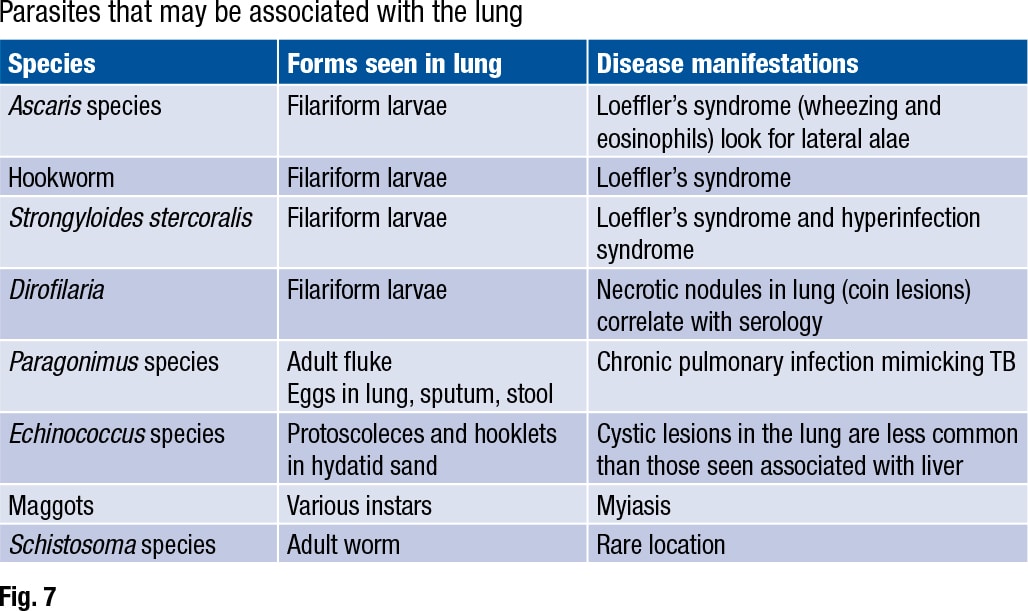
 The Strongyloides life cycle includes a migratory phase through the lung (Fig. 7). Ascaris, hookworm, and Strongyloides stercoralis are all associated with “a migratory phase through the lung, often stimulating a brisk eosinophilia, wheezing, and other respiratory symptoms,” Dr. Ribes said. This is called Loeffler’s syndrome. “Filariform larvae may be seen in bronchoalveolar lavage and sputum for all these worms, so correlate with O&P on stool for a definitive identification.”
The Strongyloides life cycle includes a migratory phase through the lung (Fig. 7). Ascaris, hookworm, and Strongyloides stercoralis are all associated with “a migratory phase through the lung, often stimulating a brisk eosinophilia, wheezing, and other respiratory symptoms,” Dr. Ribes said. This is called Loeffler’s syndrome. “Filariform larvae may be seen in bronchoalveolar lavage and sputum for all these worms, so correlate with O&P on stool for a definitive identification.”
Strongyloides rhabditiform larvae should be distinguished from hookworm rhabditiform larvae. The two are often mistaken for one another, she said, and both can appear in stool specimens. Strongyloides rhabditiform larvae are usually the diagnostic form for strongyloidiasis.
“The way we tell them apart is by looking at their mouth and at their genital primordium,” she said (Fig. 8). The Strongyloides on the left has a short buccal cavity, compared with the hookworm on the right, which has a long one. “Long is defined as greater than one-third of the total diameter of the worm itself.” The Strongyloides has a prominent genital primordium, and the hookworm, an inconspicuous one. Otherwise, the two are similar in size and shape.
Bobbi Pritt, MD, MSc, DTM&H, director of the clinical parasitology laboratory at Mayo Clinic in Minnesota, views the hookworm versus Strongyloides distinction as “a big deal.” “Strongyloides doesn’t respond to the drug that’s used for hookworm,” she said in an interview, “but Strongyloides can be life-threatening, so you want to get the treatment right.”
Gary Weil, MD, professor of medicine at Washington University School of Medicine, says the hookworm drugs they usually give are albendazole or mebendazole, “and those have very limited activity against Strongyloides. The best drug for Strongyloides is ivermectin, but it is not very good for treating hookworm infections.”
[dropcap]D[/dropcap]r. Ribes shared a case in which a 33-year-old man from Thailand was losing weight and had a chronic cough producing non-bloody sputum. Three sputa specimens tested negative for acid-fast bacilli, but the fluorescent stain displayed large fluorescent green structures that weren’t rod-shaped bacteria. When Dr. Ribes and colleagues looked at the structures under light microscopy, they saw operculated eggs with little shoulders. The abopercular ends didn’t have knobs.
 The patient’s sputum exhibited Paragonimus species eggs. For comparison, Dr. Ribes showed an image of a Paragonimus egg on the left and a Diphyllobothrium species egg on the right (Fig. 9). The eggs, she said, can be confused for one another in stool specimens. “You are never going to find Diphyllobothrium eggs in a respiratory specimen, so there knowing your life cycle is probably a good thing,” Dr. Ribes said. “But for your boards and your recredentialing, the differences are that your Diphyllobothrium eggs tend to be a little wider and a little shorter.” They also have an operculum with no shoulders, and an abopercular knob seen at that lower arrow.
The patient’s sputum exhibited Paragonimus species eggs. For comparison, Dr. Ribes showed an image of a Paragonimus egg on the left and a Diphyllobothrium species egg on the right (Fig. 9). The eggs, she said, can be confused for one another in stool specimens. “You are never going to find Diphyllobothrium eggs in a respiratory specimen, so there knowing your life cycle is probably a good thing,” Dr. Ribes said. “But for your boards and your recredentialing, the differences are that your Diphyllobothrium eggs tend to be a little wider and a little shorter.” They also have an operculum with no shoulders, and an abopercular knob seen at that lower arrow.
Paragonimus eggs, however, do have a flattened operculum that has raised shoulders. The abopercular end lacks a knob, but it may be slightly “more pointy and may be a little bit thickened,” Dr. Ribes said. “They tend to overall have a longer length distribution and are a little thinner than your Diphyllobothrium eggs.”
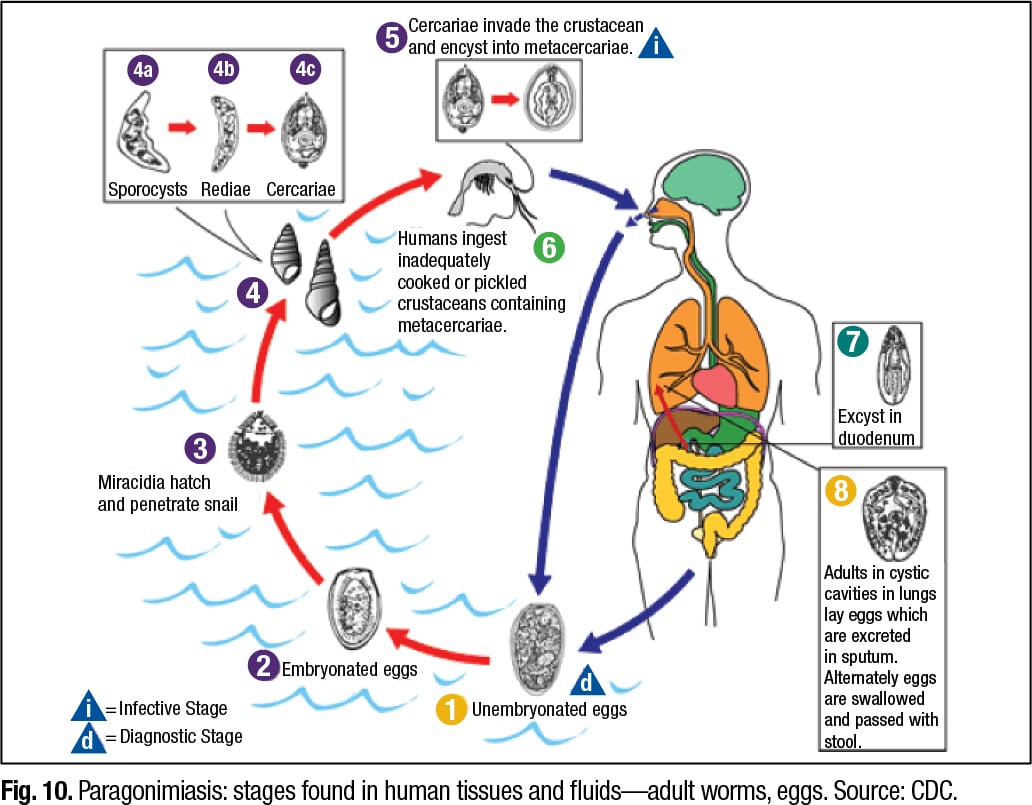
The life cycle of Paragonimus is complicated and involves snails, Dr. Ribes said. “Among the more than 10 species reported to infect humans, the most common is P. westermani, the oriental lung fluke,” says a CDC document that provides a diagram of the Paragonimus life cycle (Fig. 10).
Washington University’s Dr. Weil says paragonimiasis occurs “on every continent, but it is most important in Asia, especially in East Asia, where some 25 million people are estimated to be infected.” People become infected when they consume undercooked or uncooked crayfish or freshwater crabs. “The crustaceans are an intermediate host for the parasite, and their flesh contains larvae that infect mammals, including humans.” The larvae migrate into the lungs, evolving into adult worms over a span of months. In North America, the parasite is found only in freshwater crayfish, he says.
 Paragonimus is a hermaphrodite, Dr. Ribes said, “so both sexes in the same worm, but they are social animals. They like to have a bud, so when they first are coming into the body and are maturing in the hepatic veins, they pair up and then they migrate off to where they are going to live in perpetuity. Those that find no love may wander to other tissues like brain, where they can cause trouble such as creating seizures.”
Paragonimus is a hermaphrodite, Dr. Ribes said, “so both sexes in the same worm, but they are social animals. They like to have a bud, so when they first are coming into the body and are maturing in the hepatic veins, they pair up and then they migrate off to where they are going to live in perpetuity. Those that find no love may wander to other tissues like brain, where they can cause trouble such as creating seizures.”
“The diagnosis of paragonimiasis,” she says, “may be made either on the basis of eggs or the fluke. Eggs can be seen in the sputum, or coughed up, swallowed, and then passed in the stool. Or they can be identified in the lung on biopsy or in the fluke’s uterine horns.”
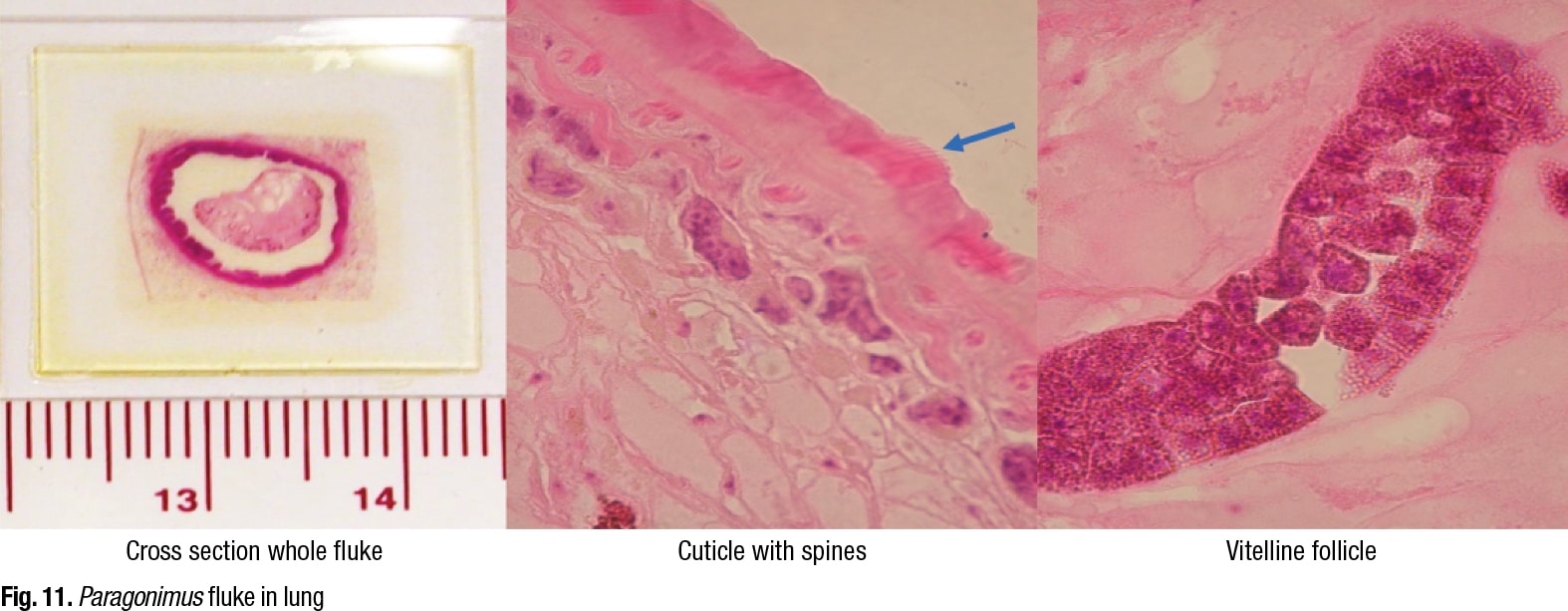
Two adult flukes generally encyst in well-developed capsules in the lung parenchyma. Adults are 4–6 mm wide and 7.5–12 mm long. The flukes have suckers, pink spines, vitellaria, ovaries, testes, and a uterus with eggs.
Dr. Ribes shared a cross section of a whole Paragonimus fluke in the lung, emphasizing that there are multiple different sections of the fluke that the tissue slice can go through (Fig. 11). She pointed to a cuticle with spines on it and to vitelline follicles. “But in this particular slice, we weren’t lucky. There is no uterus, so we don’t see any eggs.” However, “We can look for eggs within this fibrous capsule,” she said, pointing to the structure on the left side. “Or we may be able to identify eggs extravasating through the tissues into the lung parenchyma itself into the alveolar space.”
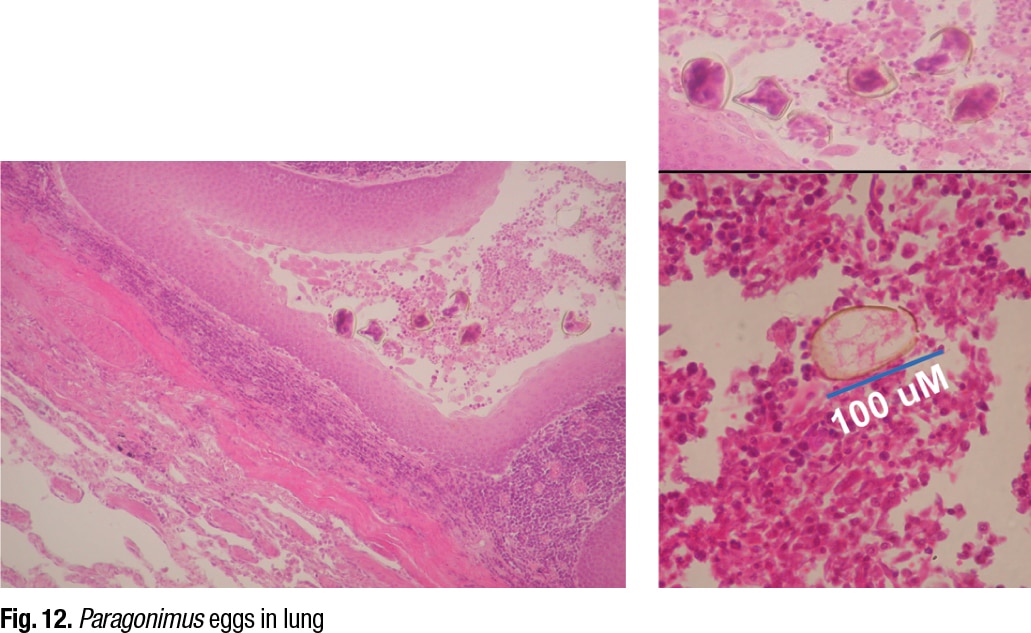
She displayed an image of Paragonimus eggs in the lung (Fig. 12), noting that the eggs are distorted because of the microtome, which is more obvious in the enlarged picture on the upper right. “We are not going to make an identification of Paragonimus based on these distorted egg structures. We might based on this,” she said, indicating the egg in the lower right image. “Here is our operculum, and there is our size distribution, location in the lung. We are good to go. We have identified this as Paragonimus species. Not necessarily anything beyond that.”
 What about other testing for Paragonimus? Dr. Weil says his colleague Peter Fischer, PhD, MS, also a professor of medicine at Washington University, developed about nine years ago an antibody test for paragonimiasis, and they shared the antigen with the CDC a year or so later (Fischer PU, et al. Am J Trop Med Hyg. 2013;88[6]:1035–1040). “Adult worms from gerbils were used to prepare an antigen for use in an antibody test,” Dr. Weil says. “Later work led to identification of a recombinant antigen that can be used instead of the native worm antigen.”
What about other testing for Paragonimus? Dr. Weil says his colleague Peter Fischer, PhD, MS, also a professor of medicine at Washington University, developed about nine years ago an antibody test for paragonimiasis, and they shared the antigen with the CDC a year or so later (Fischer PU, et al. Am J Trop Med Hyg. 2013;88[6]:1035–1040). “Adult worms from gerbils were used to prepare an antigen for use in an antibody test,” Dr. Weil says. “Later work led to identification of a recombinant antigen that can be used instead of the native worm antigen.”
The CDC, Dr. Fischer told CAP TODAY in an interview, is evaluating how the antibody test performs with its large sera collections, though the agency hasn’t published its findings yet. “Our work,” says Dr. Weil, “has focused on serology with samples from North American cases, but the test also works well with sera from patients with paragonimiasis from other regions.” He notes that the serology test is highly sensitive, but they have tested only a limited quantity of samples because human infections are infrequent in North America.
“While molecular [DNA detection] tests can be helpful,” Dr. Fischer says, “PCR of sputum or stool may provide a false-negative result because the worms cause illness long before they reach maturity and produce eggs that are shed in sputum or stool.”
Karen Lusky is a writer in Brentwood, Tenn.

