Karen Lusky
June 2017—In the CAP16 session, “Your Turn: Management of the Bleeding Patient,” Theresa Nester, MD, reminded attendees who provide transfusion medicine consultation to assess the available information before calling the clinical team: patient history, drugs, coagulation test results, and products administered so far. “Your main role is to help determine why the patient is bleeding and the most appropriate treatment,” said Dr. Nester, medical director of integrated transfusion services at Bloodworks Northwest in Seattle.

Create protocols that ‘improve communication, decrease the delivery time of blood, and provide clearly defined roles, responsibilities, and resources.’
—Theresa Nester, MD
Pathologists had an opportunity to practice that during the session. Dr. Nester and hematologist Tom DeLoughery, MD, a professor of medicine at Oregon Health & Science University, polled the audience on how they would handle various cases involving life-threatening bleeding, including potentially overwhelming hemorrhage. The speakers also provided tips for rural community hospital pathologists caring for severely bleeding trauma and obstetric patients. (They will offer the session again Oct. 10 at CAP17 in National Harbor, Md.)
Dr. Nester began with a 74-year-old man on warfarin after suffering a deep vein thrombosis who “tripped off the curb about 20 minutes ago” and now complains of headache. A head CT scan confirms he has an acute subdural hematoma. His INR is 3.5. If Kcentra (a four-factor prothrombin complex concentrate) is available, then plasma is not required. True or false?
The answer is true. Intravenous vitamin K was also indicated, Dr. Nester said, for longer-term reversal of the warfarin effect. The Kcentra package insert advises using “great caution” if the patient has had a thrombus within the last three months, she noted.
“Kcentra is the concentrate that is actually FDA-approved for use in patients on warfarin who present with life-threatening bleeding,” Dr. Nester said, adding that four-factor prothrombin complex concentrate (PCC) means it contains factors II, VII, IX, and X. If a three-factor PCC has to be given in the setting of warfarin reversal or severe bleeding, two units of plasma will have to be administered so the patient will have adequate factor VII to reverse the warfarin effect, she advised.
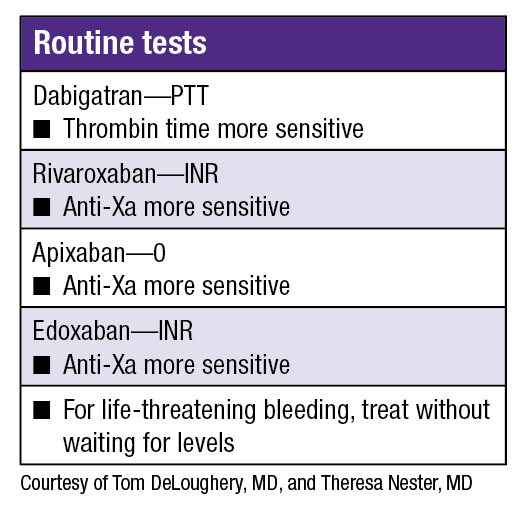
Dr. DeLoughery delved into direct oral anticoagulants (DOACs), which he noted do not require laboratory monitoring for routine use. He presented the case of a 65-year-old man on dabigatran for atrial fibrillation who presents with GI bleeding from a perforated gastric ulcer requiring urgent surgery. “The aPTT [activated partial thromboplastin time] is a useful test to determine anticoagulation due to dabigatran. True or false?” he asked. (The audience was split about 50/50 on their answers.)
“The aPTT is affected by dabigatran,” he said. “Also, given that dabigatran specifically blocks thrombin, the thrombin time is very sensitive to dabigatran effect.”
Dr. DeLoughery reviewed the coagulation tests for the DOACs which are factor Xa inhibitors. For rivaroxaban, depending on the reagent the laboratory uses, the INR can be sensitive. Also, since rivaroxaban blocks activated factor X, drug effect can be shown by using an anti-Xa assay (“heparin assay”). “If your hospital can do this testing, you can get a fast turnaround time, but if you work where it has to go to a reference laboratory, it is not going to be as helpful.”
“Even with therapeutic serum levels of apixaban, the INR and aPTT can be normal,” he cautioned. “Again, an anti-Xa assay is sensitive. With the newest agent on the market—edoxaban—the INR can be sensitive.” (See “Routine tests.”)
Returning to the case of the man on dabigatran, Dr. DeLoughery asked if the audience would recommend the patient be given a reversal agent, cryoprecipitate, prothrombin complex concentrate, or component therapy based on laboratory values.
A very specific reversal agent called idarucizumab is available for dabigatran. “It has a profound effect on reversing dabigatran action,” he said. What about bleeding with the Xa inhibitors? In a recent interview with CAP TODAY, Dr. DeLoughery said a reversal agent, andexanet alfa, currently in clinical trials may be available this year. Meanwhile, OHSU and other institutions use four-factor PCCs to reverse the effect of the Xa blockers. In his talk, Dr. DeLoughery said there will be clinical trials to test prothrombin complex concentrates as DOAC reversal agents.
Dr. DeLoughery next discussed a 47-year-old emergency room patient with alcoholic cirrhosis who is vomiting blood from esophageal varices. “Everybody is upset in the ER,” he said. The man’s hemoglobin is 8, his PT/INR is 1.9; the PTT is 42 seconds, and the platelet count, 50,000/µL. “So, what would you recommend?” he asked. “Red cells, plasma, platelets, or nothing?”
“We used to think these patients were auto-anticoagulated,” Dr. DeLoughery said. “But if you look at data, the incidence of thromboembolism actually increases as the liver disease gets worse. So why is this? Hemostasis is a balancing act. The liver not only makes procoagulants but also all the natural anticoagulants: antithrombin, protein C, protein S. And we don’t measure those readily, and we don’t have simple screening tests for these proteins. Given both pro- and anticoagulants are decreased in liver disease, hemostasis is rebalanced.” He cautioned, however, that it is rebalanced at a different state and “it’s easy to tip the balance so we can see both bleeding and thrombosis.”
As for giving the patient blood, Dr. DeLoughery cited a 2013 study of upper GI bleeding due to varices or ulcers. In the study, 461 patients were randomized to a restrictive strategy (transfusion when the hemoglobin level fell below 7 g/dL) and 460 to a liberal strategy (transfusion when the hemoglobin fell below 9 g/dL). “This is one of those trials,” he said, “you’d think, wow, why would anybody look at that?” But the results showed that aggressively giving blood in this situation was actually harmful.
Dr. DeLoughery also noted that variceal bleeding is “a mechanical phenomenon.” The balloons (varices) in the patient’s esophagus have ruptured and if you “increase the pressure in the balloon by pouring in more blood, pouring in more volume, that balloon is going to keep bleeding. It needs a mechanical fix such as your GI colleague’s sclerosing [the varices] or banding them.”
The answer in this case was to do nothing. “And that’s quite a change. Five, 10 years ago we’d have been doing this, doing that. But you know we kind of accept platelets of 50,000/mL as hemostatic; we ignore the other tests, and his hemoglobin did not meet transfusion criteria. This patient was best served by somebody on staff calling a GI consultant in to do a therapeutic endoscopy.”
Focusing on massive transfusions, Dr. Nester presented a situation in which a 73-year-old man has a ruptured abdominal aortic aneurysm. His hematocrit is 13 percent but the other coagulation laboratory test results are not yet available. “So are you going to recommend a ratio of one red cell to one plasma to one apheresis platelet; a ratio of six reds to six plasma to one apheresis platelet; a ratio of three reds to two plasma with platelets in the second round—or something else?”
Dr. Nester said there was currently no perfect answer for the situation of a bleeding patient without trauma. The definition of 1:1:1 is “actually one whole-blood-derived platelet to one unit of plasma to one unit of red cells,” she said. “And an apheresis is equivalent to about six whole blood platelets.” So if you’re using apheresis platelets, then the ratio becomes six red cells, six plasma, and a dose of apheresis platelets.
If this protocol is started, for how long should it continue? Not long, she said. The goal is to move to component therapy based on laboratory tests as quickly as possible. However, if the laboratory has not established specific protocols for an emergency hemorrhage panel, the coagulation tests may be resulted too slowly to be helpful. In that situation, the clinicians are going to want to move to something simple such as a 1:1:1 protocol, even though it may not be appropriate for non-trauma situations.
The PROMMTT (Prospective, Observational, Multicenter, Major Trauma Transfusion) study showed that trauma patients who received ratios less than 1:2 (one unit of plasma to two units of red cells) in the first six hours were three to four times more likely to die compared with those who received a 1:1 ratio (one plasma to one red cell), Dr. Nester said (Holcomb JB, et al. JAMA Surg. 2013;148[2]:127–136). “This was the first large—involving 10 U.S. level one trauma centers—observational study providing evidence that if you give plasma and platelets ‘up front’ in a big resuscitation, you have a higher chance of saving your patient.”
Then the PROPPR (Pragmatic, Randomized Optimal Platelets and Plasma Ratios) trial in trauma patients asked a more specific question, she said: whether the 1:1:1 ratio is non-inferior to a 1:1:2 (1 whole blood platelet:1 unit plasma:2 RBCs) ratio (Holcomb JB, et al. JAMA. 2015;313[5]:471–482). The randomized, controlled trial included 680 severely injured trauma patients. There was no significant difference in mortality or adverse events between the two groups. However, as a secondary outcome, the 1:1:1 group had greater proportion of hemostasis and lower mortality due to exsanguination at 24 hours.
The authors wrote, “Exsanguination, which was the predominant cause of death within the first 24 hours, was significantly decreased in the 1:1:1 group (9.2% vs 14.6% in 1:1:2 group; difference, −5.4% [95% CI, −10.4% to −0.5%]; P = .03).”
As for whether the 1:1:1 ratio pertains to a non-trauma patient, Dr. Nester cited a Canadian consensus study from 2011 (before the PROPPR trial came out), in which the expert opinion was that “if the patient has similar physiology to a trauma patient—i.e. there is evidence of shock, acidosis, and significant tissue injury—then perhaps the data in trauma does apply (Dzik WH, et al. Crit Care. 2011;15[6]:242). If not, there is insufficient evidence for the patient with a ruptured aortic aneurysm to know the correct strategy,” she said. From a coagulopathy standpoint, “most patients with ruptured aneurysm are not going to start out with a coagulation disorder unless they are on antiplatelet therapy,” she said. “The rare aneurysm patient will develop DIC [disseminated intravascular coagulation].”
“What can we take away from the trauma literature?” Dr. Nester asked. “From a high-level view, you have a ‘golden hour’ to save a patient with a ruptured aortic aneurysm.” Referring to the “lethal triad”—hypothermia, coagulopathy, acidosis—she noted that patients “will get too cold, too acidotic, too coagulopathic to save them if you don’t give them some plasma and platelets during the first part of the resuscitation.” And if the laboratory test results aren’t available for an hour, “you have missed the boat. You have to give a bolus of plasma and platelets before that time. So even in a ruptured aneurysm patient, this lethal triad idea still very much applies.”
 CAP TODAY Pathology/Laboratory Medicine/Laboratory Management
CAP TODAY Pathology/Laboratory Medicine/Laboratory Management
