HBsAg tests
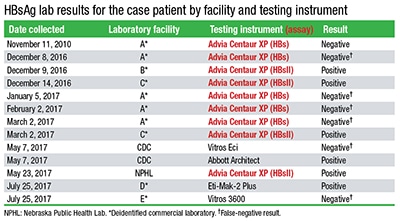
In “HBsAg tests, mutation in public health spotlight” (April 2018), you reported correctly that a patient had a false-negative hepatitis B surface antigen test performed on a Siemens Advia Centaur XP using the older HBs assay. However, the accompanying table (from MMWR Morb Mortal Wkly Rep. 2018;67:311–312) listed the Advia Centaur XPT. You published a note below the table to clarify that Lab A was in fact not using the Centaur XPT: “CAP TODAY was informed at press time that Lab A was not using the Advia Centaur XPT at the time of the false-negatives as it [the laboratory] first reported and as reflected in the table. It was using the Advia Centaur XP, which is compatible with the older HBs assay as well as the HBsII that was adapted to detect some of the common HBsAg mutations. HBsII was used at Labs B and C and at the NPHL.”
[lightbox full="https://www.captodayonline.com/2018/Portal/HBsAg.jpg"]Click Here[/lightbox]
Siemens Healthineers routinely seeks opportunities to improve its products in order to achieve its goal of clinical excellence.
Melanie Pollan, PhD, MT(ASCP)
Director, Scientific and Clinical Affairs
North America, Laboratory Diagnostics
Vera Bitcon, MS, MT(ASCP)
Director, Infectious Diseases
Global Marketing, Laboratory Diagnostics
Siemens Healthineers
Tarrytown, NY
Electronic cancer checklists
I was excited to see the article “Clearing the air for electronic cancer checklists” (May 2018) and would like to share what we at Sunquest are doing to contribute to adoption of the electronic cancer checklists.
Sunquest is continually improving the PowerPath Synoptics tool set, which is included as part of our base system. We have introduced enhancements to our synoptic worksheets editing tool based on pathologists’ feedback to improve usability, streamline the completion of required fields, and deliver real-time previews of report formatting. This new version of the Synoptics tool includes a basic rules engine to suggest specific checklists and auto-populate answers based on specimen and case information. A rules-based approach can provide more flexibility for pathologists who find customizations necessary. The team has also upgraded our conversion tool enabling us to reduce turnaround time in delivering published CAP updates to PowerPath clients for implementation.
 CAP TODAY Pathology/Laboratory Medicine/Laboratory Management
CAP TODAY Pathology/Laboratory Medicine/Laboratory Management
