
Esther Diana Rossi, MD, PhD
Daniel F. I. Kurtycz, MD
William C. Faquin, MD, PhD
 May 2018—The Milan System for Reporting Salivary Gland Cytopathology was published Jan. 31 and is an important step toward standardizing the reporting of salivary gland fine needle aspiration.1
May 2018—The Milan System for Reporting Salivary Gland Cytopathology was published Jan. 31 and is an important step toward standardizing the reporting of salivary gland fine needle aspiration.1
A large body of literature has demonstrated that FNA is an effective method for the initial evaluation of salivary gland masses, but until this year there was no uniform, widely accepted reporting system. The complexity of salivary gland cytology poses unique challenges that demand a standardized approach to communication of diagnostic information between pathologists and treating clinicians. Establishing a common reporting language promotes education and research in salivary gland cytology just as other diagnostic cytology systems have done in their respective areas of cytopathology. Creation of such reporting systems is a natural consequence of the maturation of evidence-based literature and clinical experience. Thus it is that the first atlas for salivary gland cytopathology is now available.
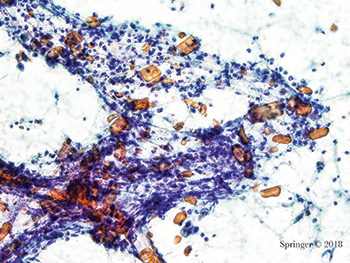
Non-neoplastic. Amylase crystals are non-birefringent crystalline structures with rectangular, needle-shaped, rhomboid, and platelike shapes. They are most commonly associated with non-neoplastic inflammatory conditions as in this case (smear, Papanicolaou stain).
The cytomorphology of a variety of salivary gland tumors has been defined over decades of practice. Unique features of a number of salivary gland lesions, including the cytomorphologic overlap of some benign neoplasms and low-grade carcinomas, have been well described. The accuracy of salivary cytology is high for the diagnosis of the most common salivary gland tumors such as pleomorphic adenoma and Warthin tumor, but the specificity for subtyping a particular neoplasm shows a wide range (48 to 94 percent) depending on tumor type. Accuracy is generally high for distinguishing benign and low-grade neoplasms from high-grade carcinomas.2–13 It is useful to remember that the nature and types of a given salivary gland lesion (neoplastic and non-neoplastic) vary depending on anatomic site. The rate of malignancy varies from 20 to 25 percent in the parotid gland, from 40 to 50 percent in the submandibular gland, and up to 80 percent in minor salivary glands of the oral cavity.
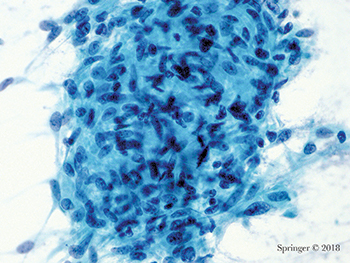
Non-neoplastic. This aspirate of granulomatous sialadenitis shows a large group of epithelioid histiocytes; an infectious agent should be excluded (smear, Papanicolaou stain).
The overall effectiveness of salivary gland FNA at a given institution depends on a number of factors, including the use of ultrasound guidance, rapid on-site evaluation, the cytologic preparation used (liquid-based, alcohol-fixed smears, and air-dried smears), and technical and diagnostic experience. Optimizing these factors enhances the diagnostic utility of salivary gland FNA. Various aspects of the lesion, such as tumor subtype, lesional heterogeneity, and presence of cystic elements, will also influence the diagnostic accuracy of the FNA.
There had been no uniform classification system for reporting salivary gland FNAs. Thus, the American Society of Cytopathology with the support of the International Academy of Cytology established an international working group, composed of more than 40 participants from 15 countries. The group consisted of cytopathologists, surgical pathologists, and head and neck surgeons. Their goal was to establish a tiered reporting system, which has now been designated the Milan System for Reporting Salivary Gland Cytopathology (MSRSGC). The name reflects the first gathering of the core working group during the 2015 annual meeting of the European Congress of Cytology in Milan, Italy.
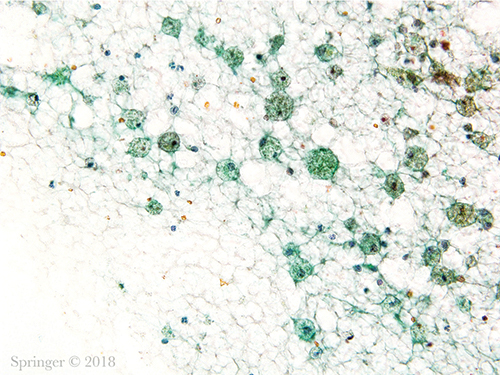
Non-diagnostic. Non-mucinous cyst contents showing histiocytes, debris, and a few inflammatory cells (smear, Papanicolaou stain).
The Milan System consists of six diagnostic categories (Table, below). It is an evidence-based system that correlates diagnostic categories with risk of malignancy and clinical management. The soft-bound atlas has 10 chapters that contain detailed descriptions of each of the six diagnostic categories of the Milan System. The atlas incorporates explanatory notes and color image FNA examples for each entity. The six diagnostic categories are non-diagnostic, non-neoplastic, atypia of undetermined significance (AUS), neoplasm subclassified into benign and salivary gland neoplasm of uncertain malignant potential (SUMP), suspicious for malignancy, and malignant.
The opening chapter of the atlas describes indications for salivary gland FNA as well as FNA technique, associated preparation methods, and implied risks of malignancy. The final three chapters are dedicated to ancillary testing for FNA, recommendations for clinical management, and histologic considerations.
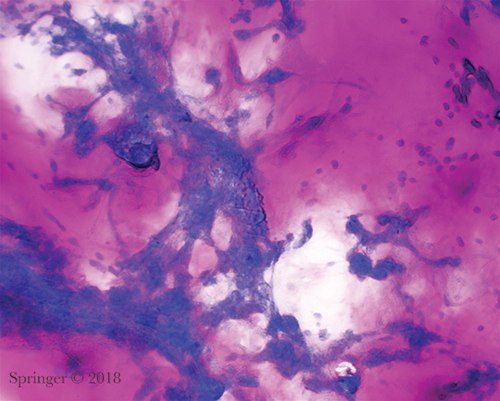
Pleomorphic adenoma. The stroma lacks the usual fibrillary character and mimics thick mucin (smear, Romanowsky stain).
The Milan System is based on key principles developed for standardized terminologies adopted for other anatomic sites, including the uterine cervix, pancreas, and urinary system. The atlas format is similar to that of The Bethesda System for Reporting Thyroid Cytopathology.14 (See article For thyroid cytopathology, the 2017 Bethesda System.) The objective of the working group was to create a user-friendly system that will promote and standardize communication between cytopathologists and treating clinicians, cyto-histological correlation, and the exchange of information among institutions.
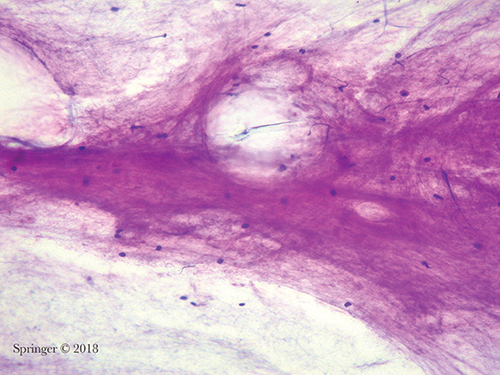
Atypia of undetermined significance. This aspirate contains abundant mucin without any epithelial cells. The differential diagnosis includes a benign mucinous cyst; however, a low-grade mucoepidermoid carcinoma cannot be excluded (smear, Romanowsky stain).
The non-diagnostic category includes samples with significant limitations, often due to scant cellularity or preservation artifact. Also included are samples characterized by nonmucinous cyst contents, aspirates of normal salivary gland tissue only, and aspirates lacking adequate material for evaluation. An online consensus survey opinion was that samples showing only normal salivary gland elements in the context of a defined clinical or radiologic mass should be deemed non-diagnostic.15 This is in keeping with the concept of the “triple test” in that cytologic, radiologic, and clinical findings should be concordant. The non-diagnostic chapter supports radiologic correlation and emphasizes the value of ultrasound guidance for its value in defining and targeting a lesion.
 CAP TODAY Pathology/Laboratory Medicine/Laboratory Management
CAP TODAY Pathology/Laboratory Medicine/Laboratory Management
