But what if the laboratory provided quantitative results? Dr. Melanson asked. “Same patient, but the methadone was sky high, greater than the reporting limit of your assay. There was EDDP there, but it was right at the cutoff, about 5 ng/mL, so both were detected but the parent compound was significantly higher. The creatinine was still normal. If you have these quantitative results in front of you and the clinician called, would you assume this patient is compliant with their medication?”
Quantitative results are better to assess whether the patient is simulating compliance, she said. “In this case, it’s highly suggestive that the patient was dropping the methadone pill directly into their urine so they would get a positive result for methadone.” The low level of EDDP showed that the patient took the prescribed methadone at some point but was currently not compliant.
Quantitative results also allow laboratories to normalize results to urine creatinine, which is helpful for monitoring THC or other drugs the clinician would like to see decrease over time. “If results are not normalized, you might see the results for THC continue to go up,” Dr. Melanson said. “But maybe the urine is getting more concentrated leading to higher results and in fact the patient has stopped using THC. If you normalize to creatinine, you can tell the difference and the clinician can more accurately determine if results are going down over time.”
With quantitative results, the laboratory can also assess whether a drug is present simply because it is an impurity in one of the drug formulations. “Morphine may have a bit of codeine, oxymorphone may have a little bit of oxycodone in it. That wouldn’t be consistent with the patient metabolizing the compound,” she said. The amount of the impurity is usually less than 0.1 percent of the parent drug or the formulation prescribed. Having quantitative results allows the laboratory to “feel pretty comfortable that this is actually due to an impurity and they have not taken an additional drug.”
Dr. Melanson argued against performing hydrolysis by presenting the disadvantages: It is time-consuming and prolongs turnaround time, chemical hydrolysis can degrade opiate and benzodiazepine drugs, and enzymatic hydrolysis can be incomplete and even reduce nordiazepam to oxazepam. “It’s better not to hydrolyze for these limitations and include the glucuronides” for more accurate assessment of opioid prescription compliance, she said.

Dr. Melanson
Dr. Melanson disagreed that hydrolysis was necessary for better sensitivity. “Assays are very good now. If we go down in the lower limit of quantitation, assays will be sensitive enough to measure both the free and glucuronidated drugs.”
She presented data showing that adding the glucuronides helped the laboratory better assess whether a patient was taking prescribed morphine. “We used to measure only morphine and the minor metabolite hydromorphone to assess compliance,” she said. Adding the glucuronides, which are present in all patients taking morphine, resulted in an increase in the positivity rate, according to the data, and improved detection of compliance (Gencheva R, et al. Clinical benefits of direct-to-definitive testing for monitoring compliance in pain management. Pain Physician, in press).
The lower limit of quantitation (LLOQ) “increases the positivity rate particularly for illicit drugs, which are concerning in all patient populations,” Dr. Melanson said. It also will help reduce the incidence of false-negatives. “You won’t be as affected by patients who have dilute urine or matrix effects.”
The data showed increased detection of illicit drugs when the laboratory used the LLOQ. “It increased the positivity rate for all combinations: heroin and fentanyl, cocaine and fentanyl, cocaine and heroin, and morphine and fentanyl. We were better able to identify these patients for the clinicians when we use the LLOQ,” Dr. Melanson said.
Brigham and Women’s laboratory leaders compromised on the points of debate and went live with their new LC-MS/MS testing panel in August 2017. “We are detecting 37 drugs and metabolites, and these were determined in conjunction with our clinicians,” given the patient population and what drugs are prescribed, Dr. Melanson said.“We went with lower cutoffs and we decided not to perform hydrolysis,” she said. “It was an additional three hours, so we added the glucuronides instead.”
Eliminating the immunoassay saved Brigham and Women’s, which already had the mass spectrometer, about $100,000 in reagents and send-out costs. The laboratory was also able to remove an analyzer that had been dedicated to those immunoassay drug screens.
The testing panel reflects further compromise between reporting quantitative and qualitative results, Dr. Melanson said. “We actually do think that qualitative results are sufficient in most cases, so the majority of new analytes are reported qualitatively.” If the results do not fit the clinical picture or seem suspicious, the laboratory “can always reflex to a quantitative result.”
The laboratory stayed with quantitative results for opioids and benzodiazepines that were already being tested in-house. “We had been doing them since 2010, so the clinicians were used to getting quantitative results,” she explained. Clinicians also preferred quantitative results for buprenorphine in particular. “It is a drug for which we see a lot of simulated compliance, and there are published papers showing that you can use the ratios of parent to metabolite as well as the naloxone to determine if the patient is compliant.”
Since there is no hydrolysis step and therefore many drugs and metabolites are reported, Dr. Melanson said the laboratory is considering systematically interpreting results for clinicians based on the patient medications prescribed at the time of collection. “We feel it’s important to help our providers interpret these,” she said, adding that the laboratory is currently studying how well clinicians are interpreting results since the implementation of the new assay.
“If we get an email from a provider, we will answer on a case-by-case basis to try to help them assess compliance,” she said. Adding a comment to some or all results may also be helpful. In an example of a patient who was prescribed clonazepam and buprenorphine, the results showed acceptable levels of the parent compound clonazepam and the metabolite 7-aminoclonazepam, but high levels of buprenorphine and very low levels of the metabolites. The patient also had very high levels of naloxone, which suggested the patient was not taking the buprenorphine as prescribed. “We might provide a comment to alert the clinician that the patient most likely dropped the buprenorphine pill directly into their urine,” she said.
Fentanyl and its metabolite, morphine, high levels of the morphine glucuronides, and hydromorphone were also detected. “All in all, this suggests they’re taking fentanyl and morphine,” Dr. Melanson said. “We might add a comment that says, ‘Suggestive of fentanyl and morphine use, prescriptions for which were not found in the medical record,’” and that those results could be from heroin use.
Drs. Melanson and Petrides plan to publish their findings on how well the clinicians are interpreting results once the study is complete.
In the debate over oral fluid versus urine as the specimen of choice, Dr. Melanson referred to the laboratory’s study, which showed that amphetamines and the heroin metabolite 6-acetylmorphine were detected at higher rates in oral fluid. She also supported oral fluid for being noninvasive, less likely to be adulterated, more effective at detecting drugs in real time, and resisting poppy products and overhydration.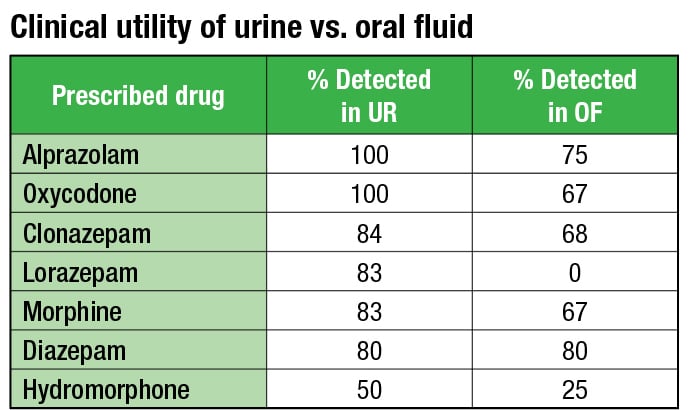
Adapted from Petrides AK, et al. Clin Chim Acta. 2018;481[6]:75–82.
Drs. Melanson and Petrides reached a compromise by reviewing patient records to determine the performance of each matrix for prescribed medications. “For prescribed medications in the study—alprazolam, oxycodone, clonazepam, lorazepam, morphine, diazepam, and hydromorphone—the detection rates were higher in urine than oral fluid,” Dr. Petrides said, adding that it therefore may be better to use urine in a pain management setting but not in an addiction setting.
They looked more closely at the heroin metabolite 6-acetylmorphine, which gets metabolized to morphine. 6-AM was better detected in oral fluid, in accordance with its biochemical properties. Their data showed that morphine did not have a preference for the matrix in which it was detected.
“What we hypothesized was that where you would find the morphine is dependent on the time of collection in relation to the time of ingestion,” Dr. Petrides said.
Plotting the oral fluid-to-urine ratio of morphine concentration revealed a time course. In oral fluid, there is a bell-shaped curve. “When you see a low result in oral fluid, it can either be due to recent use or it can be due to remote use. It’s hard to say whether if you get a positive result for 6-AM in oral fluid exactly when the patient took it,” Dr. Petrides said. “So oral fluid is the preferred matrix for 6-AM and amphetamine. But using a ratio of oral fluid-to-urine morphine could assist with interpreting 6-AM results.”
The final proposed testing algorithm settled on oral fluid as the preferred matrix for 6-AM and amphetamines, and urine for 7-aminoclonazepam, lorazepam, oxazepam, hydromorphone, oxymorphone, buprenorphine, and cocaine. More studies are needed to determine the preferred matrix for fentanyl, tramadol, MDMA/MDA, and methadone.
Dr. Petrides closed the session on a note about continued challenges from clinicians. A 45-year-old woman with advanced cervical cancer was prescribed oxycodone for pelvic pain. When a urine toxicology panel (by LC-MS/MS) revealed the presence of benzoylecgonine, a cocaine metabolite, she denied using cocaine. The patient suggested to her physician that her oral fluid contact with her dog, which had been prescribed the antibiotic clindamycin, caused her urine to test positive for cocaine. The clinician requested a test for clindamycin for the patient.
“This was a real case,” Dr. Petrides said. “It was pretty interesting to try to convince the clinical team that this could not possibly ever happen.”
Amy Carpenter Aquino is CAP TODAY senior editor.
 CAP TODAY Pathology/Laboratory Medicine/Laboratory Management
CAP TODAY Pathology/Laboratory Medicine/Laboratory Management
